

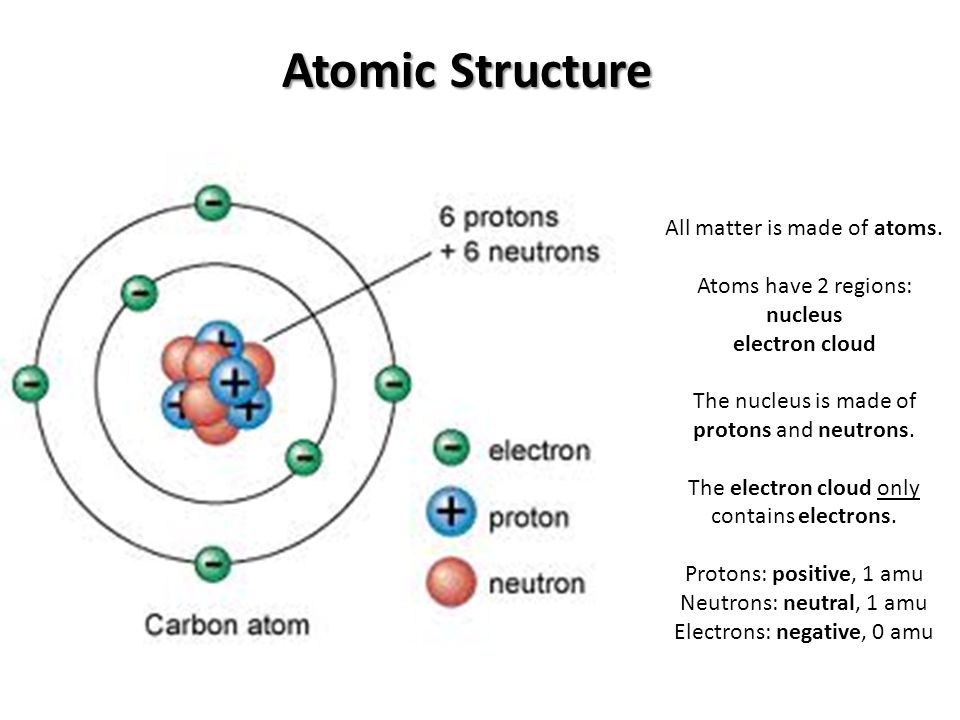
So, the sub-energy levels are 4s, 4p, 4d, and 4f. So, the sub-energy levels are 3s, 3p, and 3d. So, the sub-energy levels are 2s, and 2p. The sub-energy levels are known as s, p, d, and f. The sub-energy levels depend on the azimuthal quantum number. The most probable region of electron rotation around the nucleus is called the orbital. These sub-energy levels are also called orbital. Electron configuration of carbon atom through orbitalĪtomic energy shells are subdivided into sub-energy levels. The electron configuration of all the elements can be done through the orbital diagram. The electron configuration of an element with an atomic number greater than 18 cannot be properly determined according to the Bohr atomic model. Therefore, the order of the number of electrons in each shell of the carbon(C) atom is 2, 4.Įlectrons can be arranged correctly through orbits from elements 1 to 18. Therefore, a carbon atom will have two electrons in the first shell and four in the 2nd shell. That is, the number of electrons in carbon is 6. The atomic number is the number of electrons in that element.Ĭarbon atom electron configuration (Bohr model) Therefore, the maximum electron holding capacity in the first shell is two, the second shell is eight and the 3rd shell can have a maximum of eighteen electrons. The maximum electrons holding capacity in N orbit is 2n 2 = 2 × 4 2 = 32. The maximum electrons holding capacity in M orbit is 2n 2 = 2 × 3 2 = 18. The maximum electron holding capacity in L orbit is 2n 2 = 2 × 2 2 = 8. The maximum electron holding capacity in K orbit is 2n 2 = 2 × 1 2 = 2. The electron holding capacity of each orbit is 2n 2. K is the name of the first orbit, L is the second, M is the third, and N is the name of the fourth orbit. These circular paths are called orbit(shell). The electrons of the atom revolve around the nucleus in a certain circular path. The complete idea of the orbit is given there. Scientist Niels Bohr was the first to give an idea of the atom’s orbit.
ATOM DIAGRAM HOW TO
How to write the orbital diagram for carbon?.Electron configuration of carbon atom through orbital.Carbon atom electron atom configuration through orbit.This is in the ultraviolet part of the spectrum, so it would not be visible to us. This is the energy carried away by the photon.Ĭonverting this to joules gives E = 10.2 * 1.60 x 10 -19 J/eV = 1.632 x 10 -18 J
In dropping from the n = 2 state to the ground state the electron loses 10.2 eV worth of energy. One way to do this is to first calculate the energy of the electron in the initial and final states using the equation: What is the wavelength of the photon? Is this in the visible spectrum? When it drops to the ground state a photon is emitted. The electron in a hydrogen atom is in the n = 2 state.
If a photon with an energy equal to the energy difference between two levels is incident on an atom, the photon can be absorbed, raising the electron up to the higher level. In the hydrogen atom, with Z = 1, the energy of the emitted photon can be found using:Ītoms can also absorb photons. The photon has a smaller energy for the n=3 to n=2 transition. The smaller the energy the longer the wavelength. The photon emitted in the n=3 to n=2 transition.The photon emitted in the n=4 to n=2 transition.To conserve energy, a photon with an energy equal to the energy difference between the states will be emitted by the atom.Ĭonsider the photon emitted when an electron drops from the n=4 to the n=2 state to the photon emitted when an electron drops from n=3 to n=2. If the electron in the atom makes a transition from a particular state to a lower state, it is losing energy. The n = 1 state is known as the ground state, while higher n states are known as excited states. It's often helpful to draw a diagram showing the energy levels for the particular element you're interested in. Energy level diagrams and the hydrogen atom

